 Trauma
Trauma
Program: Trauma
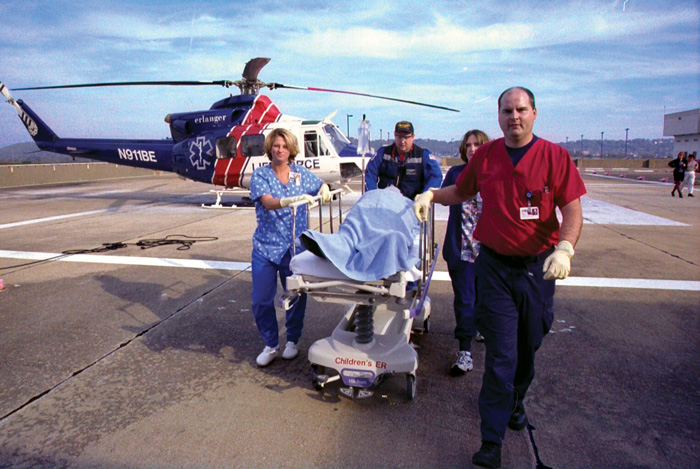
Position: UTHSC physicians operate three of the state’s six Level 1 Trauma Centers treating nearly 12,000 cases annually, which places UTHSC trauma centers among the top facilities in the nation by volume. For the past 25 years, UTHSC trauma centers have been among the world leaders in research advances in trauma care.
Memphis Leaders: Martin A. Croce, MD, UTHSC professor of general surgery, and chief, Division of Trauma and Critical Care; and Timothy C. Fabian, MD, UTHSC professor and chairman, Department of Surgery
Mission: To provide around-the-clock access to advanced life-saving care for the most severely injured patients of the state and region, while simultaneously training generations of trauma clinicians and researchers who will make future breakthroughs in trauma and critical care.
Designated Level 1 Trauma Center: 1985 – Elvis Presley Memorial Trauma Center in The Regional Medical Center at Memphis; 1987 – UT Medical Center, Knoxville; 1988 – Erlanger Health System, Chattanooga
 Current Teams: Memphis – Eight trauma surgeons and team members treat 4,500 cases annually; Knoxville – Six trauma surgeons and team members treat 3,700 cases each year; Chattanooga – Five trauma surgeons and team members treat 2,800 patients a year.
Current Teams: Memphis – Eight trauma surgeons and team members treat 4,500 cases annually; Knoxville – Six trauma surgeons and team members treat 3,700 cases each year; Chattanooga – Five trauma surgeons and team members treat 2,800 patients a year.
Core Strength: Teamwork that brings patients back from the brink on a regular basis.
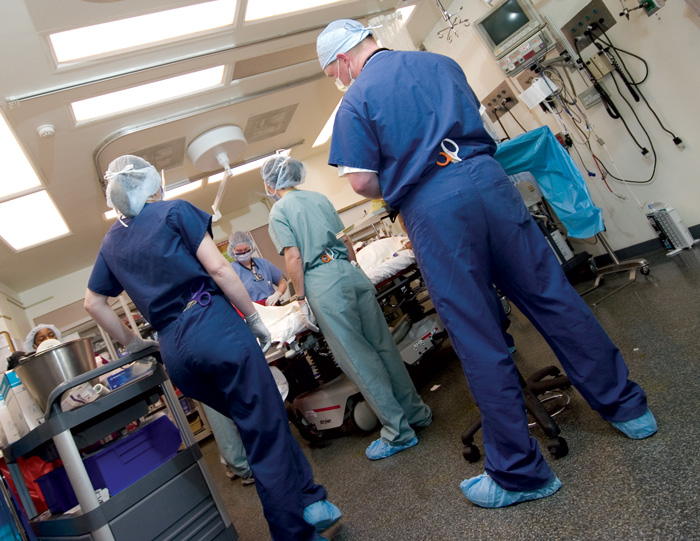
Life-Saving Assets: The state of Tennessee, using criteria developed by the American College of Surgeons Committee on Trauma (ACSCOT), designated all three UTHSC facilities as Level 1 Trauma Centers. The ACSCOT determines the criteria that separate a Level 1 Trauma Center from the typical hospital ER or a lower level trauma unit. All three UTHSC Trauma Centers include:
- Surgical capability 24/7/365 – A typical trauma team includes a general surgeon, neurosurgeon, orthopedic surgeon, radiologist, anesthesiologist, and trauma nurses.
- Clinical research conducted and published.
- Medical education offered.
- Public education promoted.
 Adult Cancer
Adult Cancer
Program: Adult Cancer
Position: The Center for Cancer Research (CCR) is the Mid-South’s only adult cancer research facility.
UT Cancer Institute (UTCI) is a partnership between UTHSC and Boston-Baskin Cancer Group that combines research and teaching capabilities with patient care from the Mid-South’s largest cancer physicians group.
Leaders: Lawrence M. Pfeffer, PhD, director, Center for Cancer Research, Muirhead Professor and vice-chair, Department of Pathology; Furhan Yunus, MD, chief medical officer, UT Cancer Institute, and interim chief, Division of Hematology and Oncology; and Guy Reed, MD, Diggs Alumni Professor and chair of the Department of Medicine
Vision: To develop a premier, patient-centered cancer center that provides integrated, cutting-edge care, and to foster research to improve cancer treatment and prevention.
Founded: CCR – 2005; Dr. Pfeffer was charged to bring together well-funded investigators from the various colleges with a general interest in cancer research to discuss being housed in the Cancer Research Building, which opened in 2007
UTCI – 2001; Partnership organized to develop a clinical infrastructure and a critical mass of clinical faculty to establish a regional comprehensive clinical cancer center.
Current Teams: CCR –15 principal investigators from medicine, dentistry and pharmacy, and some 50 other scientists
UTCI – Nearly 200 employees in 10 locations throughout Tennessee, Arkansas and Mississippi
Core Strength: CCR – “Our multidisciplinary research approach and a general openness to share resources, expertise and knowledge with others. This general philosophy of group science fits well with the modular concept of the building that has [32] laboratories without walls, which breaks down the barriers between investigators.” – Dr. Pfeffer
UTCI – Sees 60,000 patients per year with 3,000 being new. Has six multidisciplinary teams [malignant hematology, thoracic, breast, gentourinary, head and neck, and gastrointestinal oncology], providing clinical cancer care while conducting disease-specific cancer research. Conducts 40 open cancer clinical trials at any given time and trains 12 fellows annually to excel in research and patient care.
In conjunction with St. Jude Children’s Research Hospital, UTCI is a member of the National Comprehensive Cancer Network (NCCN), a not-for-profit alliance of 21 of the world’s leading cancer centers. NCCN develops clinical practice guides for the treatment of cancer; its guidelines are the world’s standard for providing cancer care.
FUTURE Plans: CCR – Recently formed specific working groups in skin, oral, brain, breast and urological cancer, as well as in experimental therapeutics to facilitate establishment of a successful cancer center. The short-term goal of these groups is to encourage interactions between basic and clinical investigators in order to enhance translational research. Some of the drugs and molecular diagnostic and prognostic tests that are presently in the laboratory stage may be brought into the clinical setting.
UTCI – Short-term goal is to develop a regional comprehensive cancer institute. Obtaining National Cancer Institute (NCI) designation as a comprehensive cancer center remains the long-term goal.
